This new breakthrough in battery technology could be a big step toward safer and more efficient energy storage, and it all comes down to how ions move inside a battery. Researchers in the US have developed a special kind of superionic polymer that may solve one of the biggest problems holding back solid-state batteries.
Inside every battery, ions travel between two electrodes during charging and discharging. The speed and ease of that movement directly affect how well the battery performs. Traditional batteries use liquid or gel electrolytes, but solid-state batteries are gaining attention because they promise better safety and higher energy density. The challenge has been finding a solid material that allows ions to move quickly enough.
That’s where this new polymer comes in. Scientists engineered a lithium salt-based material that enables ions to move incredibly fast, overcoming the usual limitations of polymer electrolytes. While polymers are flexible and easier to manufacture compared to ceramics, they’ve always struggled with slow ion transport. This new design changes that completely.
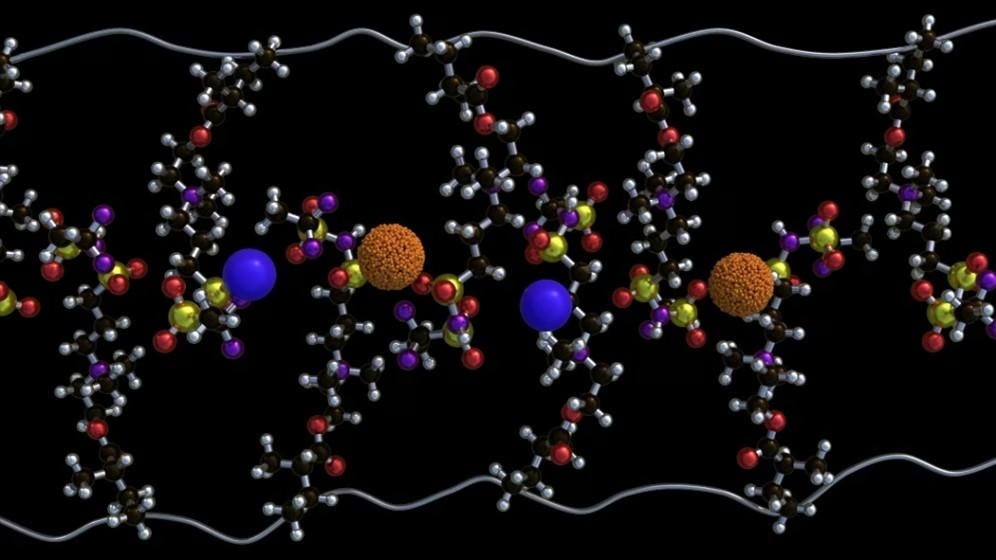
What makes this material different is how its internal structure is carefully tuned. The researchers introduced special molecular groups called zwitterions into the polymer. These groups carry both positive and negative charges, which helps create localized regions that attract and organize lithium ions.
As more of these zwitterions are added, something interesting happens. The ions start forming tiny clusters, almost like pockets within the material. Over time, these pockets connect and form continuous pathways, allowing ions to move through the polymer much more efficiently. Instead of struggling through the material, ions can now hop smoothly from one site to another.
The team found that the best performance happens when about 80 percent of the polymer structure includes these functional groups. At that point, the material forms stable channels that dramatically boost conductivity. In fact, ions can move billions of times faster compared to their surrounding environment, which is a huge leap forward.
This kind of improvement could make solid-state batteries far more practical for real-world use. It brings together the best of both worlds: the flexibility and manufacturability of polymers, combined with the high performance typically seen in ceramic materials.
Looking ahead, the researchers want to better understand exactly how this superionic behavior works at the molecular level. They’re planning to use advanced tools like supercomputing, AI-driven chemistry, and neutron scattering to observe how ions interact inside the material in real time.
If this research continues to progress, it could open the door to batteries that charge faster, last longer, and operate more safely, which would have a major impact on everything from electric vehicles to portable electronics.

